A) 8
B) 1
C) 10
D) 2
E) 9
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
What is the difference between a coordination compound and a complex ion?
Correct Answer

verified
A coordination compound genera...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Write the formula for diamminedichloroethylenediaminecobalt(III) bromide.
A) [CoCl2(en) (NH3) 2]Br
B) [CoCl2(en) (NH3) 2]Br2
C) [CoCl2(en) 2(NH3) 2]Br
D) [CoCl2(en) 2(NH3) 2]Br2
E) (NH3) 2Cl2(en) Co3Br
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The oxidation number of Co in [Co(NH3)4Cl2]Cl is +1.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ethylenediaminetetraacetic acid (EDTA) is
A) not useful as a chelating agent.
B) an effective antidote for heavy metal poisoning (e.g.,Pb2+ and Hg2+) .
C) a monodentate ligand.
D) known to form unstable complex ions with Fe3+,Hg2+,and Zn2+.
E) known to form complexes with platinum that inhibit the growth of cancerous cells.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The systematic name of the coordination compound K2[Co(H2O)2I4] is potassium diaquotetraiodocobaltate(II).
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
A complex ion and one or more counter ions are often the components of what type of compounds?
Correct Answer

verified
Coordinati...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
Octahedral complexes can exhibit geometric and optical isomerism.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands is most likely to form a high-spin octahedral complex with cobalt(II) ?
A) CN-
B) en (ethylenediamine)
C) NH3
D) CO
E) I-
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What name is given to the species that has the ability to hold the metal atom,in the complex ion,like a claw?
A) Claw complex
B) Grasp agent
C) Chelating agent
D) Clutch ligand
E) Clasp a ligand
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 4d electrons does a ground-state Mo atom have?
A) 1
B) 4
C) 3
D) 5
E) 2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
If the plane of polarization is rotated to the left then the isomer is ____________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the systematic name for [Cu(NH3) 4]Cl2?
A) dichlorotetraamminecuprate(II)
B) tetraamminecopper(II) chloride
C) copper(II) ammonium chloride
D) tetraaminocopper(II) chloride
E) tetramminecopper(II) dichloride
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following metals is capable of forming complexes spanning the largest range of oxidation numbers?
A) Cu
B) Sc
C) Fe
D) Mn
E) Ni
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many geometric isomers can the following square-planar complex have? 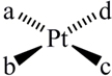
A) 1
B) 2
C) 3
D) 4
E) 6
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Molecules or ions that surround the metal in a complex ion are called ___________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures,each involving ethylene diamine and two different,unidentate ligands X and Y. 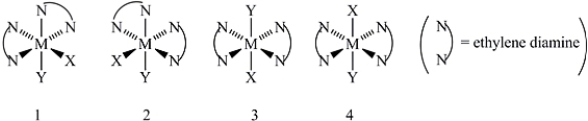 Which,if any,of the following pairs are optical isomers?
Which,if any,of the following pairs are optical isomers?
A) Structures 1 and 2
B) Structures 1 and 3
C) Structures 1 and 4
D) Structures 3 and 4
E) Structures 1,2,3,and 4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
_____________ _________ have the ability to hold the metal ion,in the complex ion,like a claw.
Correct Answer

verified
Correct Answer
verified
Short Answer
An equimolar mixture (50:50 ratio)of two enantiomers is called a(n)________ _________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species could exist as isomers?
A) [Co(H2O) 4Cl2]+
B) [Pt(NH3) Br3]-
C) [Pt(en) Cl2]
D) [Pt(NH3) 3Cl]+
E) [CuBr4]2+
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 128
Related Exams