A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of the above structures is suitable for P2I4.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of NO2- as predicted by the VSEPR theory?
A) linear
B) trigonal planar
C) bent
D) tetrahedral
E) resonant
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules has a zero dipole moment?
A) SO2
B) HCl
C) CS2
D) CO
E) Cl2O
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
The Lewis structure of formaldehyde, CH2O, is shown. Use VSEPR theory to predict the molecular geometry and the H-C-H bond angle. Outline your reasoning. 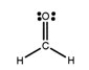
Correct Answer

verified
There are three electron groups around t...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Draw the Lewis structure of XeF4. Use this structure, in conjunction with VSEPR theory, to predict the shape of this molecule. Outline your reasoning.
Correct Answer

verified
The Lewis structure is shown below. The ...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
When resonance occurs, the bond lengths in a molecule fluctuate rapidly.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX3 will have a ______ molecular shape.
A) linear
B) bent
C) trigonal planar
D) tetrahedral
E) trigonal pyramidal
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX4 will have a ______ molecular shape.
A) bent
B) trigonal planar
C) trigonal pyramidal
D) square planar
E) tetrahedral
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX6 will have a ______ molecular shape.
A) tetrahedral
B) trigonal planar
C) trigonal bipyramidal
D) hexagonal
E) octahedral
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 109 of 109
Related Exams