B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the minimum voltage required for the electrolysis of 1.0 M NaCl in neutral solution? 2H2O + 2Cl- (1.0 M) → H2(1 atm) + Cl2(1 atm) + 2OH-(1 × 10-7 M) 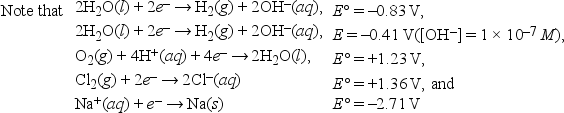
A) 2.19 V
B) 1.77 V
C) 0.41 V
D) -0.41 V
E) -1.78 V
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation using the set of smallest whole numbers coefficients. What is the sum of the coefficients? HI + HNO3 → I2 + NO (acidic solution)
A) 5
B) 7
C) 14
D) 17
E) None of these choices is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction in the lead-acid cell Pb(s) + PbO2(s) + 2H2SO4(aq) → 2PbSO4(aq) + 2H2O(l) For which E°cell = 2.04 V at 298 K. ΔG° for this reaction is
A) -3.94 × 105 kJ/mol.
B) -3.94 × 102 kJ/mol.
C) -1.97 × 105 kJ/mol.
D) -7.87 × 102 kJ/mol.
E) -7.87 × 105 kJ/mol.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the standard free-energy change for the following reaction at 25°C? (F = 96,500 C • mol-1) 2Al(s) + 3Sn4+(aq,1 M) → 2Al3+(aq,1 M) + 3Sn2+(aq,1 M) 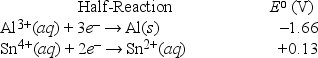
A) -1.79 kJ/mol
B) 1.79 kJ/mol
C) -1.04 × 103 kJ/mol
D) -3.45 × 102 kJ/mol
E) 5.18 × 102 kJ/mol
G) A) and E)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
If a substance is reduced, it must undergo a
A) gain of electrons.
B) loss of oxygen.
C) gain of hydrogen.
D) loss of electrons.
E) gain of oxygen.
G) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
A metal object is to be gold-plated by an electrolytic procedure using aqueous AuCl3 electrolyte. How much gold may be deposited in 3.0 min by a constant current of 10. A?
A) 6.2 × 10-3 mol
B) 9.3 × 10-3 mol
C) 1.8 × 10-2 mol
D) 3.5 × 10-5 mol
E) 1.6 × 102 mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In the electrolyte of an electrochemical cell, current is carried by electrons moving from the anode to the cathode.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is E°cell for a galvanic cell represented by the combination of the following half-reactions? 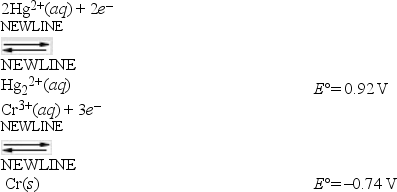
A) -0.18 V
B) 0.18 V
C) 1.28 V
D) 1.66 V
E) 2.12 V
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔG° for the following reaction? (F = 96,500 C • mol -1) I2(s) + 2Br-(aq) → 2I-(aq) + Br2(l)
Given: 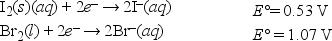
A) +104 kJ/mol
B) -104 kJ/mol
C) +309 kJ/mol
D) +52 kJ/mol
E) -52 kJ/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cell can be prepared from copper and tin. What is the E°cell for the galvanic cell that forms from the following half-reactions? 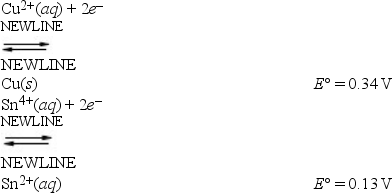
A) 0.47 V
B) 0.21 V
C) -0.21 V
D) -0.47 V
E) 0.42 V
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The redox reaction of peroxydisulfate with iodide has been used for many years as part of the iodine clock reaction. If E°cell= 1.587 V and E° of the cathode half-cell is 0.536 V, what is E° of the anode half-cell? S2O82-(aq) + 2H+ + 2I -(aq) → 2HSO4-(aq) + I2(aq)
A) -1.051 V
B) -2.123 V
C) 1.051 V
D) 2.123 V
E) None of these choices is correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element is associated with the term "galvanized"?
A) Ga
B) Zn
C) Cd
D) Hg
E) Pb
G) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What product forms at the anode during the electrolysis of molten NaBr?
A) Na+(l)
B) Na(l)
C) Br-(l)
D) Br3-(l)
E) Br2(g)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
At equilibrium E° = 0.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is not a redox reaction?
A) Al(OH) 4-(aq) + 4H+(aq) → Al3+(aq) + 4H2O(l)
B) C6H12O6(s) + 6O2(g) → 6CO2(g) + 6H2O(l)
C) Na6FeCl8(s) + 2Na(l) → 8NaCl(s) + Fe(s)
D) 2H2O2(aq) ��→ 2H2O(l) + O2(g)
E) CO2(g) + H2(g) → CO(g) + H2O(g)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The Faraday constant represents the charge of 1 mole of electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would you observe if you set up the following electrochemical cell: Ag|AgNO3(0.001 M) ||AgNO3(1 M) |Ag?
A) Electrons will flow from left to right, causing a decrease in the AgNO3 concentration in the left cell, and an increase in the AgNO3 concentration in the right cell.
B) Electrons will flow from right to left, causing an increase in the AgNO3 concentration in the left cell, and a decrease in the AgNO3 concentration in the right cell.
C) Electrons will flow from left to right, causing an increase in the AgNO3 concentration in the left cell, and a decrease in AgNO3 concentration in the right cell.
D) Electrons will flow from right to left, causing a decrease in the AgNO3 concentration in the left cell, and an increase in the AgNO3 concentration in the right cell.
E) There will be no electron flow because the reduction potential at both electrodes is the same.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much charge must pass through an electrolytic cell to reduce 0.44 mol Ca2+ ion to calcium metal?
A) 1.9 ×105 C
B) 8.5 ×104 C
C) 2.1 ×104 C
D) 4.3 ×104 C
E) 0.88 C
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the equilibrium constant at 25°C for the following reaction? (R = 8.314 J/K • mol, F = 96,500 C • mol-1) 
A) 4.1 × 1020
B) 8.2 × 1030
C) 3.3 × 1051
D) 6.7 × 1061
E) > 9.9 × 1099
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 122
Related Exams