A) Red
B) Violet
C) Orange
D) Yellow
E) Blue
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The oxidation and coordination numbers of cobalt in the compound [Co(NH3) 5Cl]Cl2 are, respectively
A) 2 and 6.
B) 2 and 8.
C) 3 and 6.
D) 3 and 8.
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
C
Correct Answer
verified
True/False
The inner transition series of elements arise from the filling of f orbitals.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species could exist as isomers?
A) [Co(H 2O) 4Cl 2] +
B) [Pt(NH 3) Br 3] −
C) [Pt(en) Cl 2]
D) [Pt(NH 3) 3Cl] +
E) None of these choices are correct.
G) B) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Chromium and manganese are among the transition elements that form several different oxides. Which of the following statements characterize these oxides?
A) As the oxidation number on the metal increases, the valence-state electronegativity increases and the oxides change from acidic to basic.
B) As the oxidation number on the metal increases, the valence-state electronegativity increases and the oxides change from basic to acidic.
C) As the oxidation number on the metal increases, the valence-state electronegativity decreases and the oxides change from acidic to basic.
D) As the oxidation number on the metal increases, the valence-state electronegativity decreases and the oxides change from basic to acidic.
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following normally acts as a bidentate ligand in complexes with transition metal ions?
A) CN −
B) EDTA 4−
C) SCN −
D) Ethylene diamine
E) Ethylene, C 2H 4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will be the strongest oxidizing agent?
A) Cr
B) Cr(II)
C) Cr(III)
D) Cr(IV)
E) Cr(VI)
G) A) and C)
Correct Answer

verified
E
Correct Answer
verified
True/False
All the actinide series of transition elements are radioactive.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron(III) forms an octahedral complex with the ligand CN-. How many unpaired electrons are in the d orbitals of iron?
A) 1
B) 3
C) 5
D) 7
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain transition element has the stable oxidation states of +2, +3, +4, +5, and +6. In which state will the element be most likely to form a covalent bond with chlorine?
A) +2
B) +3
C) +4
D) +5
E) +6
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the systematic name for [Cu(NH3) 4]Cl2.
A) Dichlorotetraamminecuprate(II)
B) Tetraamminecopper(II) chloride
C) Copper(II) ammonium chloride
D) Tetraaminocopper(II) chloride
E) None of these choices are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about lanthanides and actinides is incorrect?
A) The +3 oxidation state is the most common for both lanthanides and actinides.
B) All of the actinide elements are radioactive.
C) All of the lanthanide elements occur in nature.
D) All of the actinide elements are artificially made.
E) Due to similarities in their chemical properties, the actinide elements are not easily separated from each other.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons will there be in a high-spin octahedral complex of Fe(II) ?
A) 0
B) 2
C) 4
D) 6
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The crystal field splitting energy, Δ,
A) is larger for tetrahedral complexes than for octahedral complexes.
B) depends on the metal but not on the ligand.
C) determines the color of a complex.
D) is larger for ionic ligands like chloride than for molecular ligands like carbon monoxide, CO.
E) determines the charge of a complex.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures, each involving ethylene diamine and two different, unidentate ligands X and Y. 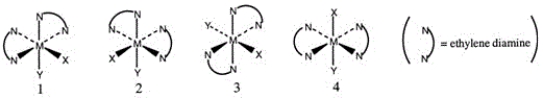 Which one of the following statements about these structures is incorrect?
Which one of the following statements about these structures is incorrect?
A) Structures 1 and 2 are optical isomers.
B) Structures 1 and 3 are optical isomers.
C) Structures 1 and 3 are different complexes.
D) Structures 1 and 4 are geometrical isomers.
E) Structures 3 and 4 are the same complex.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The conversion of the chromate ion (CrO42−) to the dichromate ion (Cr2O72−) is a redox process.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The permanganate ion (MnO4−) is a powerful reducing agent.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following has the ground state electron configuration [Ar]3d104s1?
A) In +
B) Cd 2+
C) Ag +
D) Ag
E) Cu
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Of the 3d transition series of elements, scandium has the greatest atomic radius.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the formula for diamminedichloroethylenediaminecobalt(III) bromide.
A) [CoCl 2(en) (NH 3) 2]Br
B) [CoCl 2(en) (NH 3) 2]Br 2
C) [CoCl 2(en) 2(NH 3) 2]Br
D) [CoCl 2(en) 2(NH 3) 2]Br 2
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 81
Related Exams